
- Industry news
Industry news
- Category news
Category news
- Reports
- Key trends
- Multimedia
- Journal
- Events
- Suppliers
- Home
- Industry news
Industry news
- Category news
Category news
- Reports
- Key trends
- Multimedia
- Events
- Suppliers
Key takeaways
- MIT researchers have introduced a smart pill with a biodegradable RF antenna that sends a signal confirming when a capsule has been swallowed.
- The capsule uses zinc and cellulose materials designed to break down in the stomach, while a tiny RF chip passes through the digestive system.
- The technology aims to support treatment plans for conditions requiring strict medication schedules.
Scientists from the Massachusetts Institute of Technology, US, have designed a pill capsule that contains a biodegradable radio frequency (RF) antenna. The pill reports when it has been swallowed, assisting people to take their medication on schedule.
According to the scientists behind the study, most components of the capsule break down in the stomach within a week after sending out the RF signal. The signal can be easily detected from outside the body and is said to be safe for humans. The capsule’s small RF chip leaves the body through the digestive tract.
Giovanni Traverso, the senior author of the study and an associate professor of mechanical engineering at MIT, says: “The goal is to make sure that this helps people receive the therapy they need to help maximize their health. We want to prioritize medications that, when non-adherence is present, could have a really detrimental effect for the individual.”
Ingestible solution for tracking
The MIT team aimed to create an RF-based system that would be bioresorbable to reduce the potential risk of any blockage of the gastrointestinal tract. The antenna that sends out the RF signal is made from zinc, and it is embedded in a cellulose particle.
The zinc-cellulose antenna is rolled up and placed inside a capsule together with the drug to be delivered. The outer layer of the capsule is engineered from gelatin coated with a layer of cellulose and either molybdenum or tungsten, which blocks any RF signal from being emitted.
“We chose these materials recognizing their very favorable safety profiles and also environmental compatibility,” Traverso says.
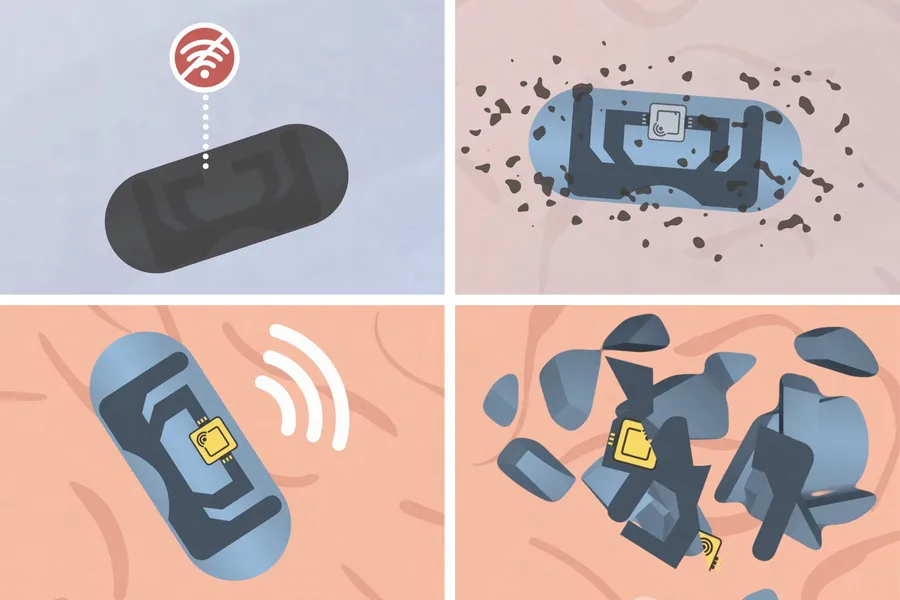 The solution aims to help ensure patients are taking their medications as prescribed (Image credit: The Massachusetts Institute of Technology).After the capsule has been swallowed, the coating breaks down, and the drug is released with the RF antenna. The antenna can then pick up the RF signal from an external receiver and, working with an RF chip about 400 by 400 micrometers, reply with a signal to confirm that the capsule was swallowed.
The solution aims to help ensure patients are taking their medications as prescribed (Image credit: The Massachusetts Institute of Technology).After the capsule has been swallowed, the coating breaks down, and the drug is released with the RF antenna. The antenna can then pick up the RF signal from an external receiver and, working with an RF chip about 400 by 400 micrometers, reply with a signal to confirm that the capsule was swallowed.
This communication is designed to take place within 10 minutes of the pill being swallowed.
Supporting treatment plans
The research, published in Nature Communications, was funded by Novo Nordisk, MIT’s Department of Mechanical Engineering, the Division of Gastroenterology at Brigham and Women’s Hospital, and the US Advanced Research Projects Agency for Health.
According to the researchers, the reporting system could help monitor transplant patients who need to take immunosuppressive drugs, or people with infections, such as HIV or tuberculosis, who need treatment for an extended period of time.
“We’ve developed systems that can stay in the body for a long time, and we know that those systems can improve adherence, but we also recognize that for certain medications, we can’t change the pill,” shares Traverso.
“The question becomes: What else can we do to help the person and help their health care providers ensure that they’re receiving the medication?”
Tests in an animal model confirmed that the RF signal was transmitted from inside the stomach and could be read by an external receiver at a distance up to two feet away. The researchers now plan to do further preclinical studies and are looking into testing the system in humans.
Mehmet Girayhan Say, an MIT research scientist and lead author of the paper, says: “The components are designed to break down over days using materials with well-established safety profiles, such as zinc and cellulose, which are already widely used in medicine.”
“Our goal is to avoid long-term accumulation while enabling reliable confirmation that a pill was taken, and longer-term safety will continue to be evaluated as the technology moves toward clinical use.”














